How Electricity Conducts Through Liquids
Electricity and liquids are deadly combination that people avoid. The process of an electric current passing through a liquid is called electrolysis. Learn more about how electricity conducts through liquids with this brief guide.
What Are Conductive Liquids Called?
Conductive liquids are known as electrolytes. Electrolytes are substances that contain free ions and can conduct electricity when dissolved in water or another solvent. There are two main types of electrolytes:
- Strong Electrolytes: These are substances that completely dissociate into ions in solution, resulting in high conductivity. Examples include salts like sodium chloride (table salt), acids like hydrochloric acid, and bases like sodium hydroxide.
- Weak Electrolytes: These partially dissociate into ions in solution, leading to lower conductivity compared to strong electrolytes. Examples include acetic acid (found in vinegar) and ammonia.
Electrolytes play a crucial role in various industrial processes, biological functions, and everyday applications. For instance, they are essential in batteries, where they facilitate the flow of electric current, and in the human body, where they help regulate nerve and muscle function.
Water
Water can be conductive but isn’t always. Distilled water contains no ions and is not conductive. Pure water contains very few ions, so it’s a poor electricity conductor. Tap water often contains salt or other impurities which add ions to the water, making it conductive.
This is why it’s unsafe to touch plugs or outlets with hands wet from tap water. The electricity will move through the liquid and can electrocute the person.
Electrolysis
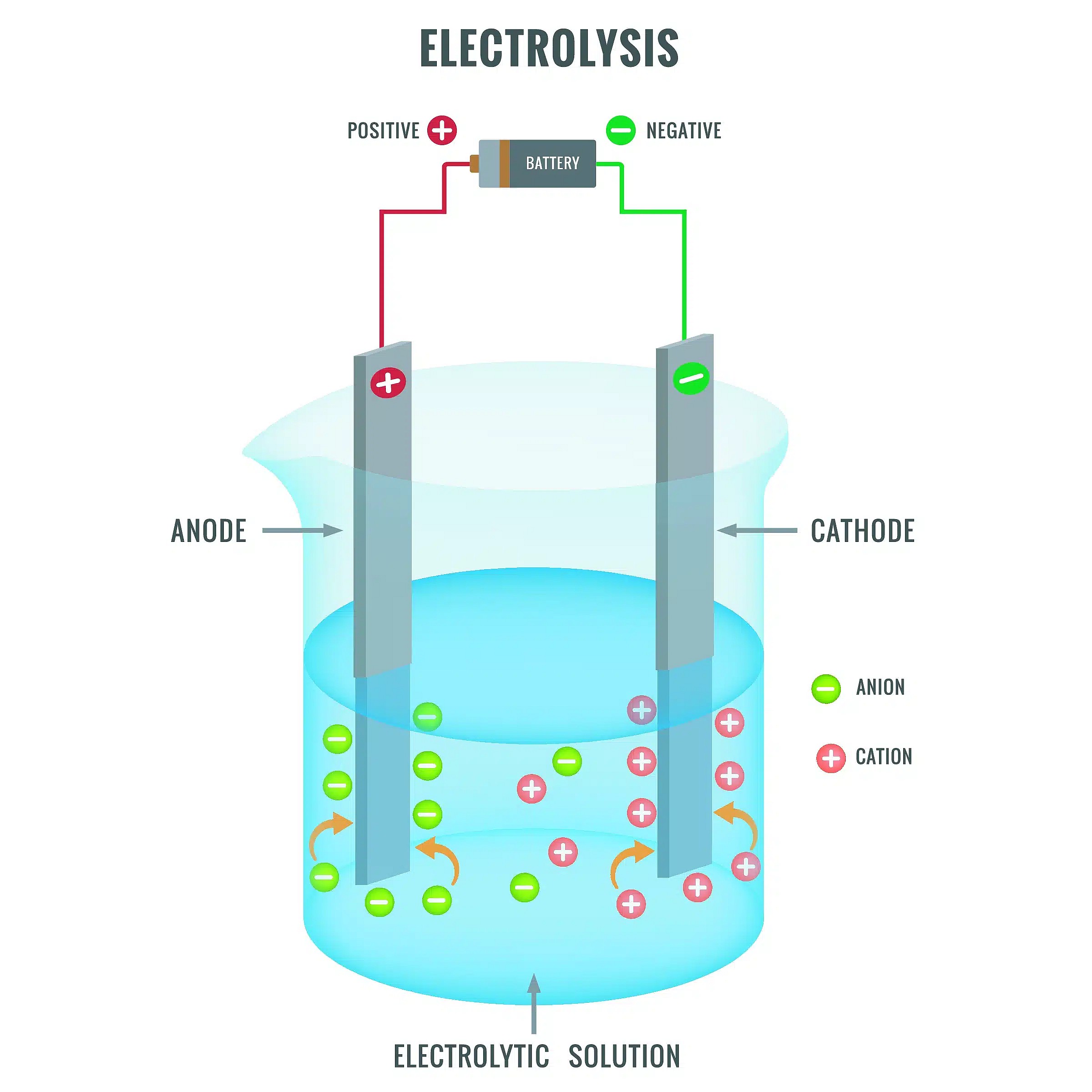
Electrolysis is a chemical process that uses an electric current to drive a non-spontaneous chemical reaction. This process is widely used in various industrial and scientific applications. During electrolysis, an electric current is passed through an electrolyte, causing the ions in the electrolyte to move towards the electrodes, where they undergo chemical reactions.
How Electrolysis Works
- Electrolyte: The substance that conducts electricity and undergoes chemical changes during electrolysis. It can be a liquid or a solution containing ions.
- Electrodes: Two conductive materials, typically made of metal or graphite, are placed in the electrolyte. The positive electrode is called the anode, and the negative electrode is called the cathode.
- Electric Current: A power source, such as a battery or power supply, provides the electric current that drives the electrolysis process.
When the electric current flows through the electrolyte, it causes the positive ions (cations) to move towards the cathode, where they gain electrons (reduction), and the negative ions (anions) to move towards the anode, where they lose electrons (oxidation). This movement of ions and the resulting chemical reactions are the basis of electrolysis.
Applications of Electrolysis
Electrolysis has numerous practical applications, including:
- Electroplating: Coating a metal object with a thin layer of another metal to improve its appearance, corrosion resistance, or other properties.
- Electrorefining: Purifying metals by removing impurities through electrolysis.
- Water Electrolysis: Splitting water into hydrogen and oxygen gases, which can be used as clean energy sources.
- Electrolysis in Water Treatment: Removing contaminants from water by breaking them down into less harmful substances.
Importance of Electrolysis
Electrolysis is essential in various industries, including manufacturing, energy production, and environmental protection. It enables the production of high-purity metals, the generation of hydrogen fuel, and the treatment of wastewater, among other applications. Understanding the principles and applications of electrolysis is crucial for advancing technology and improving industrial processes.
Non-Conducting Liquids
Liquids that are non-conducting don’t allow electricity to flow through them. These liquids have little to no ions in them. Electricity is conducted through liquids via ions, and without them, electricity cannot move through the liquid.
The conductivity of liquids can vary, and they become more conductive the more ions there are. Electrolysis is a process done intentionally to help coat metals, such as silver plating, with another substance. It’s also used to extract or purify some metals—such as refining copper by electrolysis, extracting aluminum.
For more information on the dangers of electricity passing through liquids or for professional electrical consultancy services contact Dreiym Engineering today.